 
Join my 6000+ subscribers on my YouTube Channel for new A Level Chemistry video lessons every week. Please LIKE this video and SHARE it with your friends! 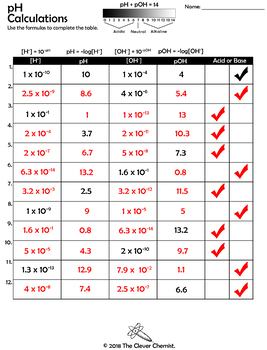
Topic: Ionic Equilibria, Physical Chemistry, A Level Chemistry, Singaporeīack to other previous Physical Chemistry Video Lessons.įound this A Level Chemistry video useful? When dealing with non-standard temperatures, we have to compare H + and OH - concentrations to deduce the nature of solution: The concept that a solution is acidic when pH is less than 7, neutral when pH is 7, and alkaline when pH is greater than 7 is only true at 25 oC or 298K. The reason why students might find this confusing is because we tend to use pH to deduce whether a solution is acidic, neutral or alkaline. This means for pure water, = will always be true and water will always be neutral regardless of temperature. It is not possible to change the nature of water by changing its temperature.Īt any temperature, regardless of the extent of dissociation, the concentration of H + and OH - will always be the same. Deduce nature of water at different temperatures Hence pH of water will be less than 7 at temperatures above 298K, and greater than 7 at temperatures below 298K.ĭoes this mean that water becomes acidic when temperature is above 298K, and water becomes alkaline when temperature is below 298K? 4. In this way you get the pH of an acid, base, salt or another chemical compound. In the simplest case the input water is pure water (H 2 O). This means H + concentration will be greater than 10 -7 mol dm -3, and the corresponding pH will be less than 7. Addition of Chemicals (pH Calculator) The reaction module (button Reac) allows the addition of chemicals to any aqueous solution (input water). Since the forward reaction is favoured, more H + and OH - is formed and their concentrations will increase. When there is an increase in temperature, the position of equilibrium will shift right to favour endothermic reaction to absorb excess heat. Recall that auto-ionisation is endothermic. Deduce pH of water when temperature is above 298K When there is a change in temperature, K w, H + concentration and therefore pH will change. Since concentration of H + and OH - in pure water are the same, we can calculate pH of water to be 7 via the following working:Īt first glance this seems hardly surprising, but pH of water is 7 only when K w = 10 -14 at 25 oC. We also need to know that auto-ionisation of water is an endothermic process. It has a given value of 10 -14 mol 2 dm -6 at 25 oC or 298K. The equilibrium constant for this dissociation is known as the ionic product of water, K w, where This process is known as auto-ionisation of water and the concentration of H + and OH - are equal since water will dissociate to give equimolar amounts of H + and OH. Pure water will undergo acid-base reaction with itself to a very small extent to form H + and OH. In this JC2 webinar we want to learn how to calculate the pH of water and deduce its nature at different temperatures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
